| Citation: | Sayyar Ali Shah, Nekmat Ullah, Shakoor Muhammad, Noor Zeb Khan. ENHANCING FRACTIONAL MODEL FOR DENGUE TRANSMISSION INCORPORATING VERTICAL TRANSMISSION AND VECTOR CONTROL[J]. Journal of Applied Analysis & Computation, 2026, 16(4): 1822-1842. doi: 10.11948/20250235 |
ENHANCING FRACTIONAL MODEL FOR DENGUE TRANSMISSION INCORPORATING VERTICAL TRANSMISSION AND VECTOR CONTROL
-
Abstract
Dengue fever continues to be a major global health concern, particularly in tropical and subtropical regions. This research develops an updated fractional-order dengue transmission model that incorporates vector control methods with vertical transmission in mosquitoes. Memory effects are considered in the model formulation, which are examined using the Caputo fractional derivative. Through vertical transmission, Infected mosquitoes transmit the virus to their offspring, which keeps the infection active when humans are not available as hosts. Spraying insecticides together with biological control measures is part of vector control efforts that help assess how well the disease prevalence decreases. The Banach fixed-point theorem provides a basis to prove existence and uniqueness solutions. The mathematical model uses the basic reproduction number ($ \mathscr{R}_0$) to determine disease persistence, while stability conditions depend on equilibrium points. The investigation of local stability uses fractional-order stability theory together with a suitable Lyapunov function to establish global stability. The disease dynamics display changes through numerical results where fractional order variations combine with vector control measures. The simulation data demonstrates that vertical disease transmission functions as a crucial factor for maintaining dengue outbreaks, but strong vector control programs can significantly reduce spreading rates. The presented model functions as a foundation to establish optimal preventive strategies against dengue fever. The subsequent part of this work investigates optimal control models.
-
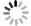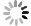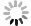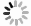
-
References
[1] A. Abidemi and O. J. Peter, An optimal control model for dengue dynamics with asymptomatic, isolation, and vigilant compartments, Decision Analytics Journal, 2024, 10, 100413. doi: 10.1016/j.dajour.2024.100413 [2] T. S. Asgarian, H. Vatandoost, A. A. Hanafi-Bojd and F. Nikpoor, Worldwide status of insecticide resistance of Aedes aegypti and Ae. albopictus, vectors of arboviruses of Chikungunya, Dengue, Zika and Yellow Fever, Journal of Arthropod-Borne Diseases, 2023, 17(1), 1. [3] A. Atangana and C. Nwaigwe, Theoretical analysis and second-order approximation of solution of fractal-fractional differential equations with Mittag-Leffler kernel, Mathematical and Computer Modelling of Dynamical Systems, 2024, 30(1), 814–839. doi: 10.1080/13873954.2024.2417720 [4] A. Bakhet, S. Hussain and M. Zayed, On fractional operators involving the incomplete Mittag-Leffler matrix function and its applications, Symmetry, 2024, 16(8), 963. doi: 10.3390/sym16080963 [5] P. Bedi, A. Khan, A. Kumar and T. Abdeljawad, Computational study of fractional-order vector borne diseases model, Fractals, 2022, 30(05), 2240149. doi: 10.1142/S0218348X22401491 [6] S. Bounouiga, B. Basti and N. Benhamidouche, Mathematical exploration of malaria transmission dynamics: Insights from fractional models and numerical simulation, Advanced Theory and Simulations, 2400630. [7] U. A. Danbaba, M. D. Aloko and A. M. Ayinde, Mathematical modeling of mosquito borne diseases with vertical transmissions as applied to Dengue, International Journal of Mathematical Sciences and Optimization: Theory and Applications, 2024, 10(3), 31–56. [8] C. S. Darby, K. M. Featherston, J. Lin and A. W. Franz, Detection of La Crosse virus in situ and in individual progeny to assess the vertical transmission potential in Aedes albopictus and Aedes aegypti, Insects, 2023, 14(7), 601. doi: 10.3390/insects14070601 [9] M. Farman, A. Ahmad, A. Akgül, M. U. Saleem, M. Rizwan and M. O. Ahmad, A mathematical analysis and simulation for Zika virus model with time fractional derivative, Mathematical Methods in the Applied Sciences, 2024, 47(13), 11135–11146. doi: 10.1002/mma.6891 [10] M. Farhan, Z. Shah, R. Jan and S. Islam, A fractional modeling approach of Buruli ulcer in Possum mammals, Physica Scripta, 2023, 98(6), 065219. doi: 10.1088/1402-4896/acd27d [11] S. Fischer, M. S. De Majo, C. Di Battista and R. E. Campos, Effects of temperature and humidity on the survival and hatching response of diapausing and non-diapausing Aedes aegypti eggs, Journal of Insect Physiology, 2025, 161, 104726. doi: 10.1016/j.jinsphys.2024.104726 [12] R. Gurgel-Gonçalves, W. K. D. Oliveira and J. Croda, The greatest dengue epidemic in Brazil: Surveillance, prevention, and control, Revista da Sociedade Brasileira de Medicina Tropical, 2024, 57, e00203–2024. [13] R. Gurgel-Gonçalves, W. K. D. Oliveira and J. Croda, The greatest dengue epidemic in Brazil: Surveillance, prevention, and control, Revista da Sociedade Brasileira de Medicina Tropical, 2024, 57, e00203–2024. [14] S. Halstead, Recent advances in understanding dengue, F1000Research, 2019, 8. DOI: 10.12688/f1000research.19197.1. [15] R. Jan, S. Boulaaras, A. Alharbi and N. N. Abdul Razak, Fractional-calculus analysis of the dynamics of a vector-borne infection with preventive measures, Fractal & Fractional, 2024, 8(12). [16] J. T. Lim, S. Bansal, C. S. Chong, B. Dickens, Y. Ng, L. Deng, C. Lee, L. Y. Tan, G. Chain, P. Ma and S. Sim, Efficacy of Wolbachia-mediated sterility to reduce the incidence of dengue: A synthetic control study in Singapore, The Lancet Microbe, 2024, 5(5), e422-e432. doi: 10.1016/S2666-5247(23)00397-X [17] Mamenun, Y. Koesmaryono, A. Sopaheluwakan, R. Hidayati, B. D. Dasanto and R. Aryati, Spatiotemporal characterization of dengue incidence and its correlation to climate parameters in Indonesia, Insects, 2024. 15(5), 366. doi: 10.3390/insects15050366 [18] O. Man, A. Kraay, R. Thomas, J. Trostle, G. O. Lee, C. Robbins, A. C. Morrison, J. Coloma and J. N. Eisenberg, Characterizing dengue transmission in rural areas: A systematic review, PLoS Neglected Tropical Diseases, 2023, 17(6), e0011333. doi: 10.1371/journal.pntd.0011333 [19] M. Meena and M. Purohit, Mathematical analysis using fractional operator to study the dynamics of dengue fever, Physica Scripta, 2024, 99(9), 095206. doi: 10.1088/1402-4896/ad671b [20] M. Z. Meetei, S. Zafar, A. A. Zaagan, A. M. Mahnashi and M. Idrees, Dengue transmission dynamics: A fractional-order approach with compartmental modeling, Fractal and Fractional, 2024, 8(4), 207. doi: 10.3390/fractalfract8040207 [21] A. Nabti and B. Ghanbari, Global stability analysis of a fractional SVEIR epidemic model, Mathematical Methods in the Applied Sciences, 2021, 44(11), 8577–8597. doi: 10.1002/mma.7285 [22] P. A. Naik, J. Zu and K. M. Owolabi, Global dynamics of a fractional order model for the transmission of HIV epidemic with optimal control, Chaos, Solitons & Fractals, 2020, 138, 109826. [23] A. J. Peterson, R. A. Hall, J. J. Harrison, J. Hobson-Peters and L. E. Hugo, Unleashing nature's allies: Comparing the vertical transmission dynamics of insect-specific and vertebrate-infecting flaviviruses in mosquitoes, Viruses, 2024, 16(9), 1499. doi: 10.3390/v16091499 [24] Y. Rachmawati, S. Ekawardhani, N. Fauziah, L. Faridah and K. Watanabe, Potential way to develop dengue virus detection in aedes larvae as an alternative for dengue active surveillance: A literature review, Tropical Medicine and Infectious Disease, 2024, 9(3), 60. doi: 10.3390/tropicalmed9030060 [25] G. Rahman, M. Samraiz, C. Yildiz, T. Abdeljawad, M. A. Alqudah and A. Mukheimer, New generalized results for modified Atangana-Baleanu fractional derivatives and integral operators, European Journal of Pure and Applied Mathematics, 2025, 18(1), 5697–5697. doi: 10.29020/nybg.ejpam.v18i1.5697 [26] M. Riaz, Z. A. Khan, S. Ahmad and A. A. Ateya, Fractional-order dynamics in epidemic disease modeling with advanced perspectives of fractional calculus, Fractal and Fractional, 2024, 8(5), 291. doi: 10.3390/fractalfract8050291 [27] D. Roiz, P. A. Pontifes, F. Jourdain, C. Diagne, B. Leroy, A. C. Vaissière, M. J. Tolsá-García, J. M. Salles, F. Simard and F. Courchamp, The rising global economic costs of invasive Aedes mosquitoes and Aedes-borne diseases, Science of the Total Environment, 2024, 933, 173054. doi: 10.1016/j.scitotenv.2024.173054 [28] N. A. Samsudin, H. Othman, C. S. Siau and Z. I. I. Zaini, Exploring community needs in combating aedes mosquitoes and dengue fever: A study with urban community in the recurrent hotspot area, BMC Public Health, 2024, 24(1), 1651. doi: 10.1186/s12889-024-18965-1 [29] Z. Shah, N. Ullah, R. Jan, M. H. Alshehri, N. Vrinceanu, E. Antonescu and M. Farhan, Existence and sensitivity analysis of a Caputo-Fabirizo fractional order vector-borne disease model, European Journal of Pure and Applied Mathematics, 2025, 18(2), 5687–5687. doi: 10.29020/nybg.ejpam.v18i2.5687 [30] A. Wilder-Smith, TAK-003 dengue vaccine as a new tool to mitigate dengue in countries with a high disease burden, The Lancet Global Health, 2024, 12(2), e179-e180. doi: 10.1016/S2214-109X(23)00590-9 [31] N. Uemura, K. Itokawa, O. Komagata and S. Kasai, Recent advances in the study of knockdown resistance mutations in Aedes mosquitoes with a focus on several remarkable mutations, Current Opinion in Insect Science, 2024, 63, 101178. doi: 10.1016/j.cois.2024.101178 [32] N. Ullah, Z. Shah, R. Jan, N. Vrinceanu, M. Farhan and E. Antonescu, Modeling the non-integer dynamics of a vector-borne infection with nonlocal and nonsingular kernel, Scientific Reports, 2025, 15(1), 6262. doi: 10.1038/s41598-025-90182-1 [33] M. Usman, M. Abbas, S. H. Khan and A. Omame, Analysis of a fractional-order model for dengue transmission dynamics with quarantine and vaccination measures, Scientific Reports, 2024, 14(1), 11954. doi: 10.1038/s41598-024-62767-9 [34] K. A. Venkatesan, Optimal control strategies for dengue fever transmission using Atangana-Baleanu fractional order models. [35] G. H. Wang, A. Hoffmann and J. Champer, Gene drive and symbiont technologies for control of mosquito-borne diseases, Annual Review of Entomology, 2024, 70. [36] Y. Wang, J. Xie and J. Zhao, Mathematical analysis of a dynamic model of epidemic influenced by super-spreaders, International Journal of Mathematical Analysis, 2024, 18(1), 21–36. doi: 10.12988/ijma.2024.912547 [37] H. M. Wanjala, M. Okongo and J. Ochwach, Mathematical model of the impact of home-based care on contagious respiratory illness under optimal conditions, Jambura Journal of Biomathematics (JJBM), 2024, 5(2), 83–94. doi: 10.37905/jjbm.v5i2.27611 [38] T. Wanjiru, W. Bulimo, S. Langat, J. Kinyua, N. Odemba, S. Yalwala, D. Oullo, R. Ochieng, F. Ngere, G. Kerich and J. Ambale, Vertical transmission of Dengue virus type-3 and metagenomic virome profiles of Aedes aegypti mosquitoes collected in Kisumu, Kenya. medRxiv, 2024, 2024, 11. [39] World Health Organization, Report of the sixth meeting of the WHO diagnostic technical advisory group for neglected tropical diseases, Geneva, Switzerland, 2024, 14–15. [40] W. Wu, J. Zhou, Z. Li and X. Tan, The effect of time delay on the dynamics of a fractional-order epidemic model, Advances in Continuous and Discrete Models, 2025, 2025(1), 9. doi: 10.1186/s13662-025-03868-1 -
-
-
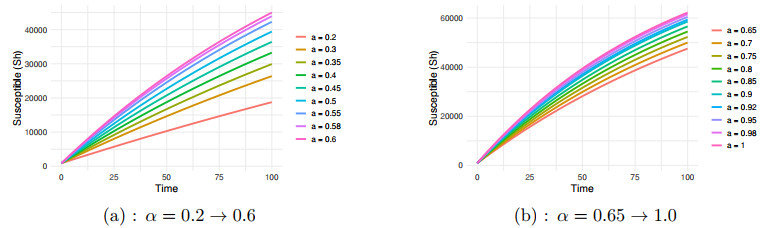
Figure 1.
$ S_h $ $ \alpha=0.2 \to 1.0 $ -
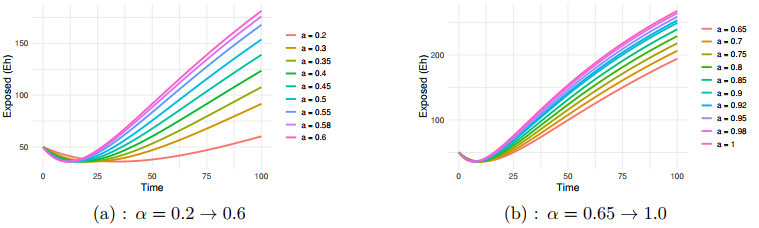
Figure 2.
$ E_h $ $ \alpha=0.2 \to 1.0 $ -
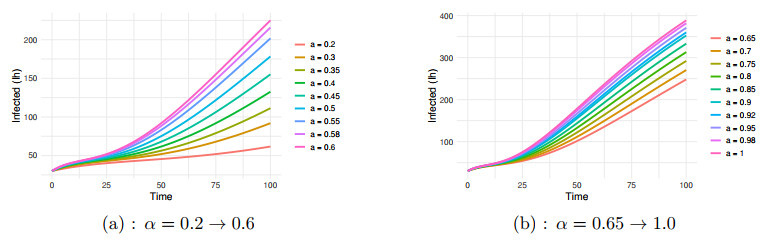
Figure 3.
$ I_h $ $ \alpha=0.2 \to 1.0 $ -
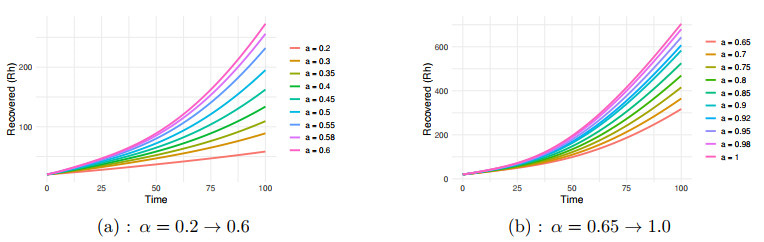
Figure 4.
$ R_h $ $ \alpha=0.2 \to 1.0 $ -
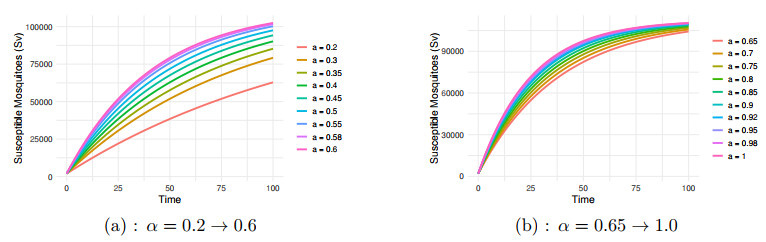
Figure 5.
$ S_v $ $ \alpha=0.2 \to 1.0 $ -
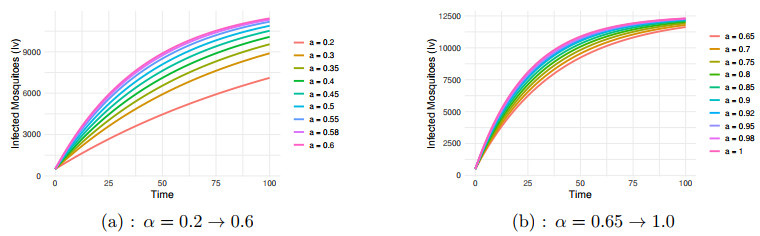
Figure 6.
$ I_v $ $ \alpha=0.2 \to 1.0 $ -
Figure 7.
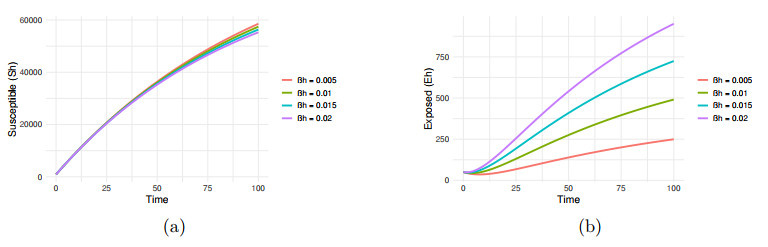
Impact of transmission rate
$ \beta_h=0.005, 0.01, 0.015, 0.02 $ $ S_h $ $ E_h $ -
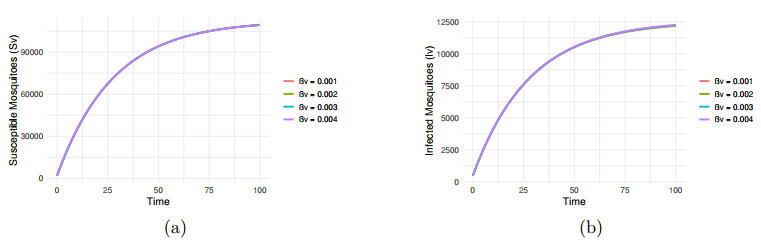
Figure 8.
Impact of transmission rate
$ \beta_v=0.001, 0.002, 0.003, 0.004 $ -
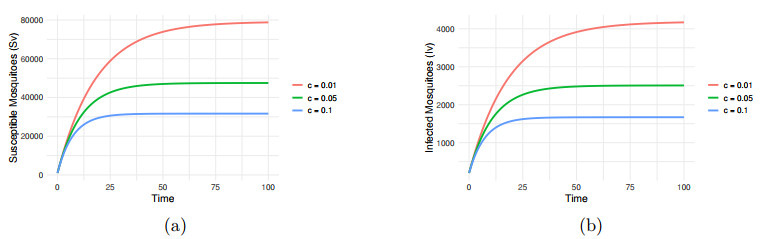
Figure 9.
Impact of vector control
$ c=0.01, 0.05, 0.1 $ -
Figure 10.
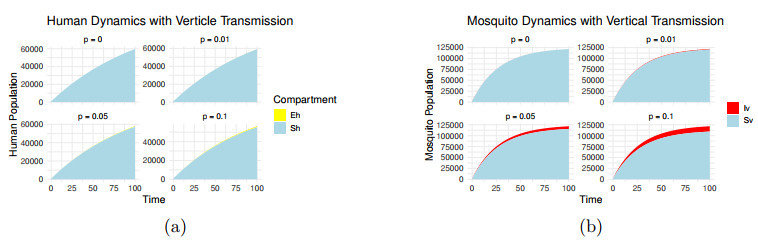
Impact of VT rate
$ p=0, 0.01, 0.05, 0.1 $ -
Figure 11.
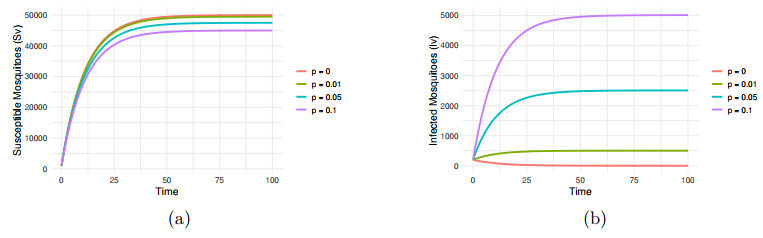
Impact of VT rate
$ p=0, 0.01, 0.05, 0.1 $



 DownLoad:
DownLoad: